DKA is an acute, major life-threatening complication of Diabetes characterized by hyperglycemia, ketoacidosis, and ketonuria. It occurs when there is absolute or relative insulin deficiency, which inhibits glucose entry into the cell. The liver then rapidly produces ketones as an alternative source of fuel, causing it to accumulate and make the blood acidic (anion gap metabolic acidosis).
DKA is predominantly seen in T1DM and is often a presenting problem in newly diagnosed patients. Intercurrent illness commonly causes DKA in established T1DM patients, as these patients are unable to adjust their insulin dose appropriately to compensate for the increased stress and insulin requirements.
Approximately 5-8 episodes of DKA per 1000 people with diabetes occur annually. DKA has a mortality rate of approximately 1%.
Diagnostic criteria for DKA
- Acidemia: Venous Blood pH <7.3 or HCO3- <15.0mmol
- Hyperglycemia: Blood Glucose >11mmol/L, Know Diabetic
- Ketonemia or significant ketonuria: ≥3.0mmol/L or 2+ on urinalysis respectively
Some variants of DKA
| Variant | Presentation | Patient History |
|---|---|---|
| Euglycemic DKA (EDKA) | Acidosis, Ketonemia, normal blood glucose | SGLTi, Pregnancy, Alcoholism, Glycogen storage disease, Very low-calorie diet, Severe liver disease |
| Ketosis-prone Diabetes | Ketoacidosis in individuals without anti-GAD and anti-islet cell antibodies. Genotypically idiopathic TIDM but phenotypically T2DM. | African American, Asian, Sub-Saharan Africa, African Carribeans. Middle-aged and Obese. |
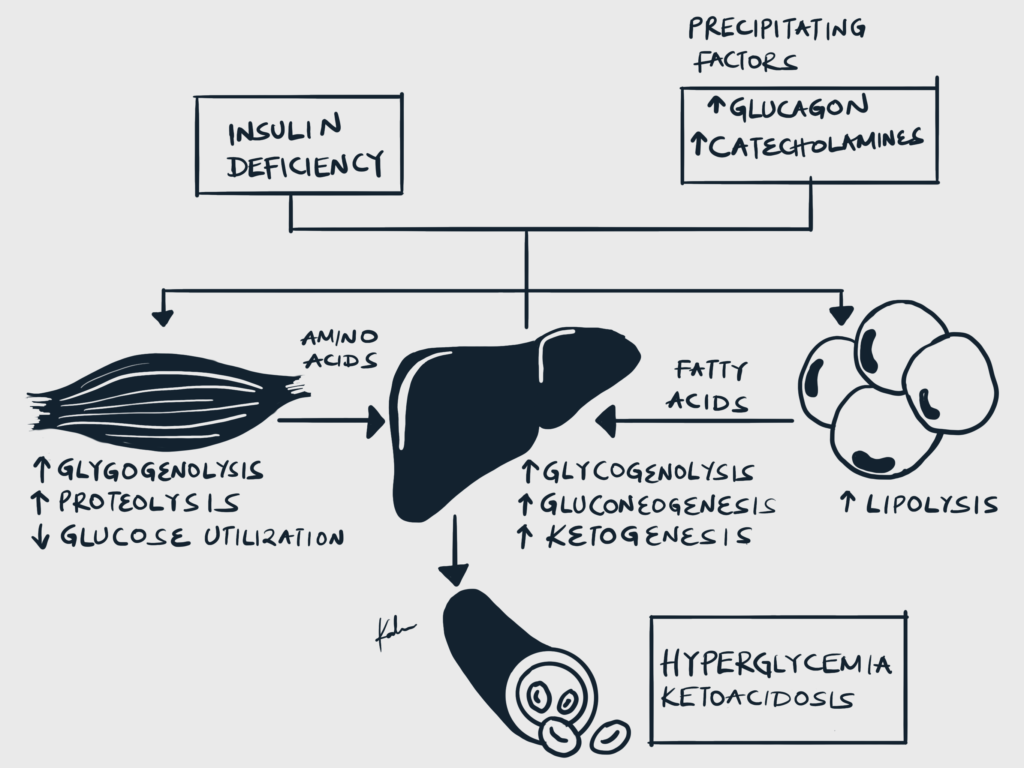
- Precipitating factors
- Infection – commonly UTI and Pneumonia
- Ischemia – myocardial infarction and stroke
- Insulin deficiency – non-compliance, treatment failure, and inability to afford treatment
- Surgery
- Trauma and Burns
- Pancreatitis
- Alcohol use
- Drugs
- Glucocorticoids
- Cocaine
- Pathophysiology
- Insulin deficiency and increased counter-regulatory hormones cause inhibition of glycolysis and increased gluconeogenesis and glycolysis → Hyperglycemia
- Insulin deficiency increases lipolysis through disinhibition of Hormone-sensitive lipase (HSL) → Lipolysis and generation of Free Fatty Acids → Ketogenesis
- Osmotic diuresis and hypovolemia
- Hyperglycemia causes hyperosmolality and osmotic diuresis, which leads to volume depletion and hypovolemia
- Metabolic acidosis with increased anion gap
- Non-esterified fatty acids (NEFAs) generated from lipolysis are converted into ketones in the liver (acetoacetate, Beta-hydroxybutyrate)
- These ketones are acidic, and bicarbonate is consumed as it buffers them.
- This causes metabolic acidosis with an elevated anion gap
- Intracellular potassium deficit
- Hyperglycemic hyperosmolality shifts potassium along with water from the intracellular to the extracellular space
- Shifted potassium in the extracellular space is lost through urine
- Insulin is absent in DKA and can therefore not promote intracellular potassium uptake
- Total body potassium deficit develops, although serum potassium may be normal or paradoxically elevated
- Signs and Symptoms
- Abdominal pain due to ketosis and delayed gastric emptying
- Nausea and vomiting
- Polyuria
- Polydipsia
- Fruity breath
- Palpitations
- Malaise
- Fatigue
- Reduced appetite
- Loss of consciousness
- Tachypnea – Kussmaul breathing
- Signs of dehydration
- Dry skin and mucous membranes
- Reduced skin turgor
- Hypotension
- Tachycardia
- Confusion
- Ill-looking patient
- Differentials
- Hyperosmolar hyperglycaemic state (HHS)
- Alcoholic ketaoacidosis
- Lactic acidosis
- Differentials for anion gap metabolic acidosis (CUTMUDPILERS)
- Carbon Monoxide and cyanide poisoning
- Alcohol (ethanol and methanol)
- Toluene
- Uraemia
- DKA
- Propylene glycol, Paraldehyde, Paracetamol
- Iron tablets, Isoniazid
- Lactic acidosis
- Ethylene glycol
- Renal failure
- Salicylates, sepsis, starvation
- Differentials for hyperglycemia and hypovolemia
- Sepsis
- Acute pancreatitis
- Differentials for altered mental status
- Hypoglycemia
- Hepatic encephalopathy
- Uremic encephalopathy
- Wernicke encephalopathy
- Investigations
- Blood Glass Analysis (best initial test)
- High-anion gap metabolic acidosis (pH <7.3)
- Random blood glucose
- Hyperglycemia (≥ 14mmol/L)
- HbA1C to determine glycemic control
- Urinalysis with dipstick
- Ketonuria
- Complete blood count
- Leukocytosis
- Left shift suggests infection
- Electrolytes
- Hyponatremia: due to osmotic diuresis or dilution as hyperglycemia shifts intracellular fluid into the extracellular space
- Hyperkalemia: insulin deficiency and acidemia shift potassium to the extracellular space. Note that total body potassium is, however, depleted and may need to be replaced when therapy is started with insulin.
- Hypomagnesemia: due to osmotic diuresis
- Urea and Creatinine
- Elevated urea and creatinine suggest AKI secondary to volume depletion secondary to osmotic diuresis
- Blood culture to rule out infection
- Hydroxybutyrate
- Elevated
- Chest X-ray to rule out pneumonia
- Electrocardiogram to rule out myocardial infarction (patients with diabetes may present with silent MI)
- Blood Glass Analysis (best initial test)
- Treatment
- Restore volume deficit through fluid replacement
- Normal saline is preferred
- Resolve hyperglycemia and acidosis using insulin
- Infusion starts at 0.1 U/kg/hour
- Once blood glucose is < 14 mmol/L, an infusion of 10% dextrose can be started at 125 mls/h in addition to the normal saline regimen
- Stop short-acting insulin and resume long-acting insulin once hyperglycemia and ketosis have resolved
- Correct electrolyte abnormalities
- Ensure K+ is > 3.3 mEq/L before administering insulin
- Treat the precipitating cause
- Additional measures
- Foley catheterization if anuric after 3 hours or incontinent
- Nasogastric tube if obtunded or persistent vomiting
- Thromboprophylaxis with Low-Molecular-Weight Heparin
- Central venous line for pregnant women, renal or cardiac failure, septic shock, severe DKA or other severe comorbidities
- EKG monitoring if severe of potassium infusion rate is > 20 mmol/hour
- Arrange a review by a diabetes specialist nurse before discharge
- Restore volume deficit through fluid replacement
- Criteria for resolution of DKA
- Glucose <11.1 mmol/L
- PLUS at least 2 of the following:
- Venous pH > 7.3
- Serum bicarbonate ≥ 15 mEq/L
- Anion gap ≤ 12 mEq/L
- Blod ketones < 0.6 mmol/L
- Complications of DKA
- Gastric stasis
- Venous thrombembolism due to dehydration, immobility, and hypervoagulable state
- Cerebral edema
- Common in children due to rapid shifts in osmolarity during treatment
- Arrhythmias secondary to severe hypokalemia and or acidosis
- Heart failure
- Acute respiratory distress syndrome
- Respiratory failure
- Mucormycosis
- Hypoglycemia due to inadequate monitoring of glucose levels during insulin therapy
- Hypokalemia
Grading the Severity of DKA
| Arterial pH | Serum bicarbonate | Anion gap | Mental Status | |
|---|---|---|---|---|
| Mild | >7.24 | 15-18 mmol/L | >10 mEq/l | Alert |
| Moderate | 7.0-7.24 | 10-15 mmol/L | >12 mEq/l | Alert or drowsy |
| Severe | <7.0 | <10 mmol/L | >12 mEq/l | Stuporous |
Example of a fluid replacement regimen for a non-hypotensive patient (SBP > 90 mmHg)
| Fluid | Volume |
|---|---|
| 0.9% sodium chloride 1L | 1000ml over 1st hour |
| 0.9% sodium chloride 1L with potassium chloride | 1000ml over next 2 hours |
| 0.9% sodium chloride 1L with potassium chloride | 1000ml over the next 2 hours |
| 0.9% sodium chloride 1L with potassium chloride | 1000ml over the next 4 hours |
| 0.9% sodium chloride 1L with potassium chloride | 1000ml over the next 4 hours |
| 0.9% sodium chloride 1L with potassium chloride | 1000ml over the next 6 hours |
Example of a potassium replacement regimen
| Serum K+ | Amount of KCl to add per litre of IV fluid |
|---|---|
| >5.5 | Nil |
| 3.5-5.5 | 40 mmol |
| <3.5 | Stop insulin. Seek help from the HDU/ICU for higher doses |
